It was announced yesterday that the first every image of a black hole was taken. This is an achievement for the scientific and specifically astrophysical community.
Astrophysics
Growing Cancer Cells & Coronal Mass Ejections Are Visually Similar
Image
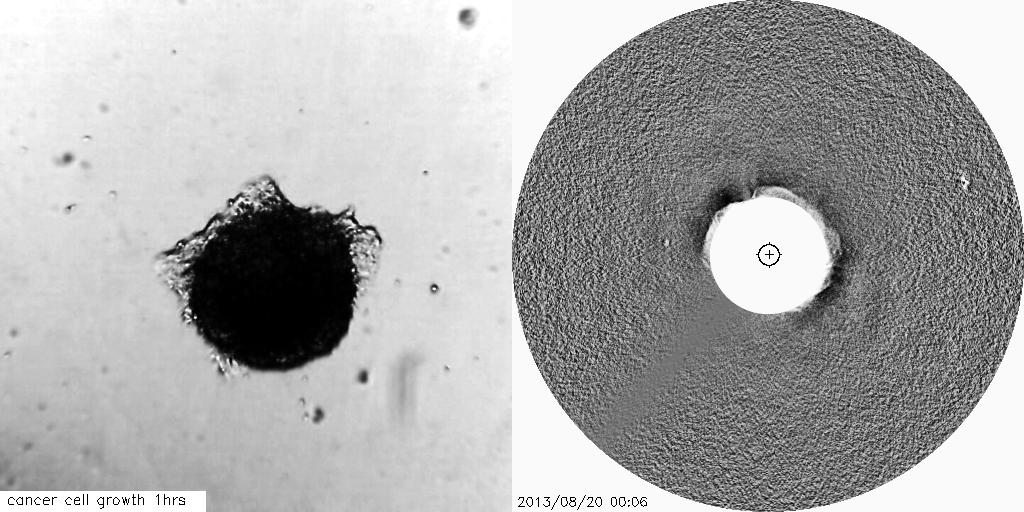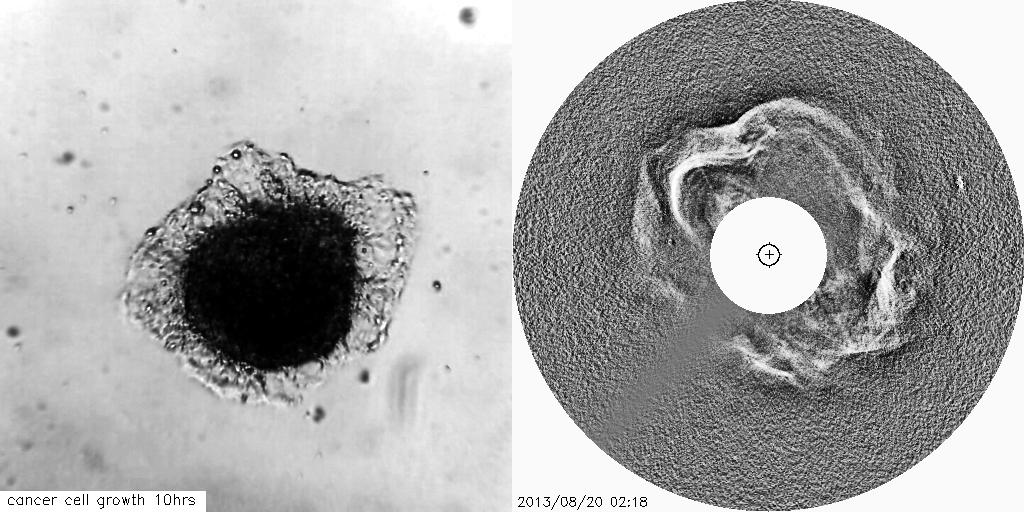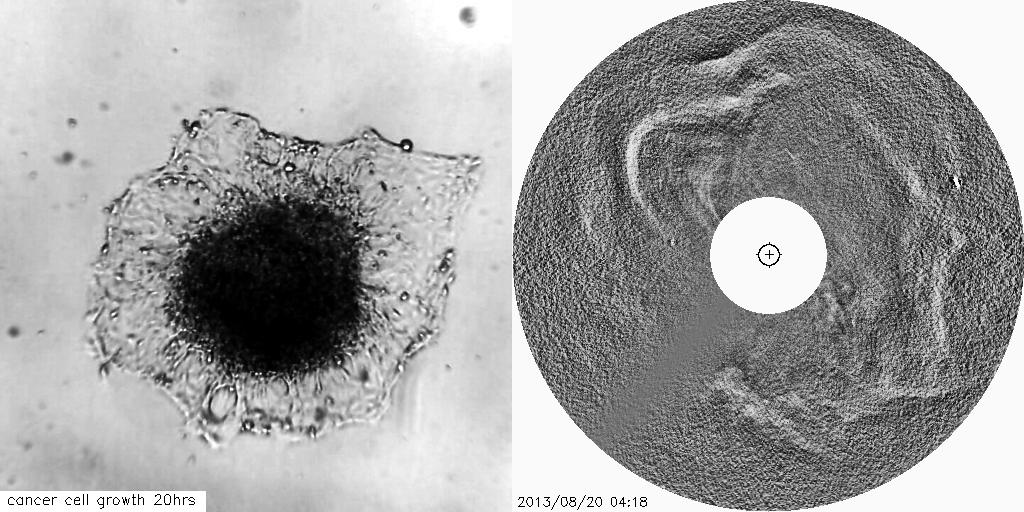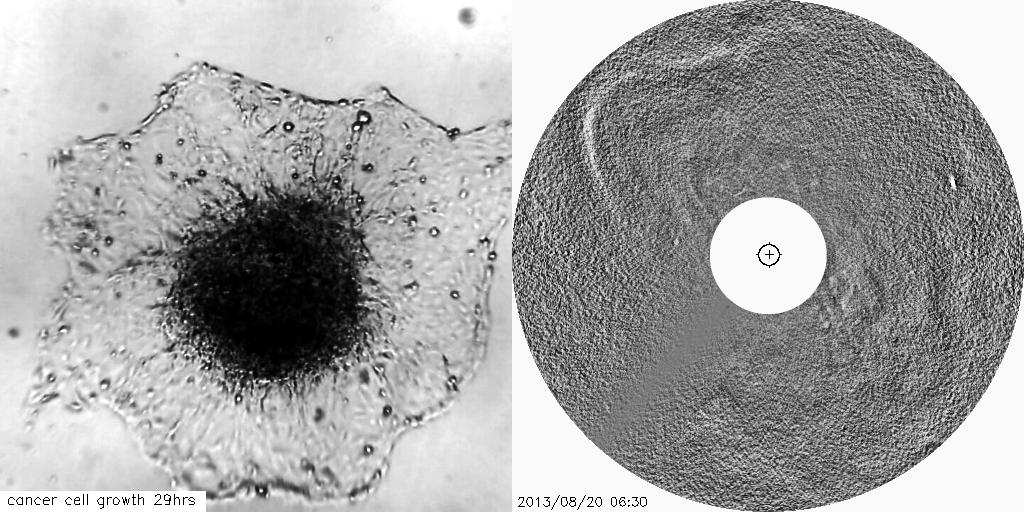
Using solar and astronomical image processing techniques and mathematical theory we have developed novel image analysis techniques to track the growth of tumours and other cell-based structures, which can be crucial for advancements of therapeutics to treat diseases such as cancers.
The visual similarities between coronal mass ejections and the outward invasion of cells from a tumour spheroid are striking. This study aims to help us better understand changes in the tissue architecture which are associated with tumour growth.
Nearly Naked Black Hole
VideoCredit: NRAO/AUI/NSF, NASA/Goddard Space Flight Center and the Advanced Visualization Laboratoy at the National Center for Supercomputing Applications, NASA/Goddard Space Flight Center/CI Lab
Vortex formation using a free jet
StandardIn a previous post I shared with you an image of milk vortices created in a hot coffee cup. Today I share with you a demonstration of vortex formation using a free jet of air of a speed higher than that (stagnant) surrounding it.
This is one case of fluid instability called the Kelvin-Helmholtz instabilities (KHI) which occurs when two fluids have different velocities. A very popular and practical example of it are sea surface waves which are created due to the velocity difference between the water and wind.
More accurately, the shear force applied by one fluid (air in this case) on the other (water) creates a shear stress in the other fluid which if greater than the surface tension of the second fluid results in the KHI.
It is also common in clouds and on gas planets like Saturn and Jupiter as the following image of Saturn show.
Image credit: NASA/JPL/Space Science Institute

You must be logged in to post a comment.